the core of our corporate values
Zylox-Tonbridge Completed Patient Enrollment for ZENFLEX® Peripheral Stent System Clinical Trial in the EU
December 18, 2023. Zylox-Tonbridge (2190. HK, "the Company"), one of the leading vascular device companies in China, announced the completion of the total enrollment of 100 patients in the post-market clinical follow-up trial for ZENFLEX®Peripheral Stent System in the European Union (EU). The study intends to evaluate the clinical performance of ZENFLEX® as well as to seek CE mark approval under EU MDR, enabling itself to better provide patients with high-quality products and services.
The trial is a prospective, multi-centre, single-arm study to evaluate the safety and efficacy of bare metal stent-assisted percutaneous transluminal angioplasty (PTA) in the treatment of superficial femoral and/or proximal popliteal artery (P1) lesions in patients with symptomatic peripheral artery. The study was led by Prof. Dr. Dierk Scheinert, a renowned international leader in peripheral intervention from the University Hospital of Leipzig, as a scientific coordinating investigator, with the participation of several clinical physicians at five sites in the EU.
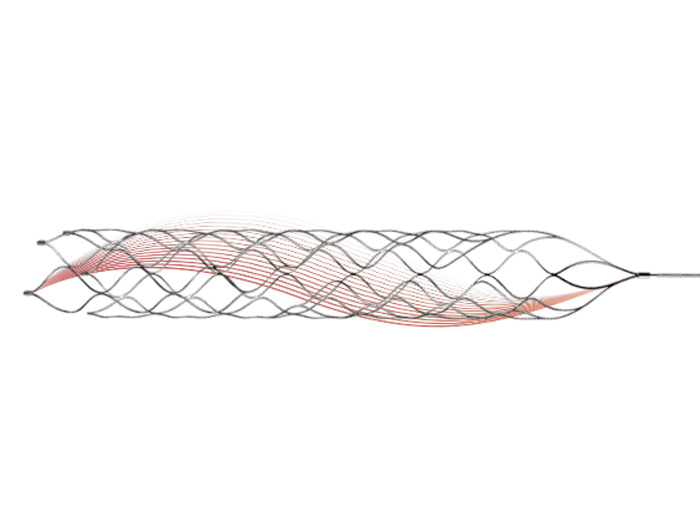
ZENFLEX® has several outstanding features. Its open helical structure and segmented micromesh design provide excellent flexibility and high radial force, ensuring immediate and continuous wall apposition. Additionally, the controlled delivery system allows ZENFLEX® more convenient to release precisely and quickly. With its innovative design and high quality, ZENFLEX® has been recognized and welcomed by local physicians since its launch in the EU in 2017.
In 2023, Tonbridge medical group has made great progress in overseas markets. The neuro-medical device companies' products were successfully commercialized in several developed overseas markets. Leveraging its strong R&D capabilities and advanced manufacturing platforms, Zylox-Tonbridge also provided OEM services to local brands in some emerging markets.
Looking forward, Zylox-Tonbridge plans to continue investing in the international markets, providing patients worldwide with more innovative neurological medical devices, peripheral medical devices and solutions.