the core of our corporate values
Zylox-Tonbridge’s ZYLOX® Phoenix Peripheral Detachable Fibered Coil Embolization System Receives NMPA Marketing Approval
Zylox-Tonbridge Medical Technology Co., Ltd. (2190.HK, hereinafter referred to as "Zylox-Tonbridge" or the "Company") announced that its ZYLOX® Phoenix Peripheral Detachable Fibered Coil Embolization System (hereinafter referred to as "ZYLOX® Phoenix"), a product independently developed by the Company, has received marketing approval from the National Medical Products Administration (the “NMPA”).
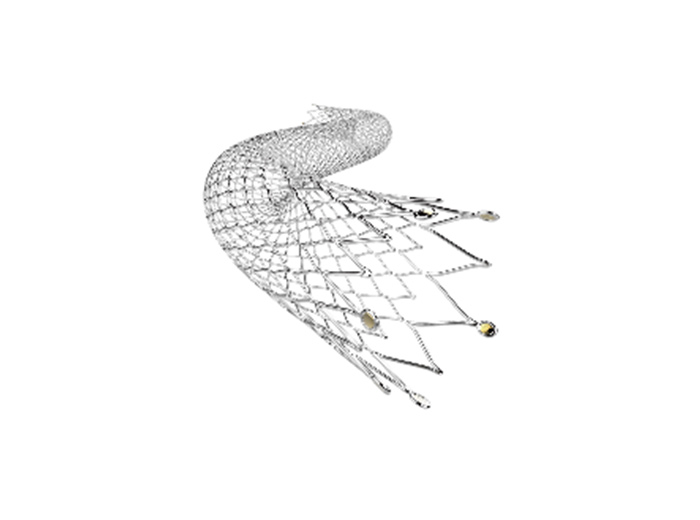
ZYLOX® Phoenix is a proprietary medical device for the minimally invasive interventional treatment of peripheral arterial embolism. The clinical application of coils for vascular embolization has been prevalent in the field of peripheral vascular intervention. For example,, endoleak management of abdominal aortic aneurysms, embolization treatment of visceral aneurysms, hemoptysis and arteriovenous fistula are important clinical applications, and their safety and efficacy have been internationally recognized. Currently, the Chinese market for peripheral vascular interventional devices is still dominated by imported brands. The launch of ZYLOX® Phoenix can effectively fill the gap in the field of domestically developed arterial embolization devices, providing physicians and patients with high-quality and affordable products and solutions.
Based on the existing embolization coil products, the ZYLOX® Phoenix optimizes and upgrades the interlocking arm detaching structure, which improves the cornering ability of the embolization coils and reduces the risk of premature detaching. Moreover, this product specifically adds polypropylene anti-unwinding wire, which further address the technical challenge of premature unwinding of embolization coils. Compared with similar products currently available in the market, ZYLOX® Phoenix is available in a wider range of length and diameter specifications, providing more options to meet the needs of various vascular embolization scenarios.
The pre-marketing clinical registration trial of the ZYLOX® Phoenix was led by Professor Zhao Jichun from West China Hospital of Sichuan University. A total of 101 patients were enrolled from 14 top clinical trial centers across the PRC were enrolled, and the immediate post-operative target vessel subtotal occlusion rate reached 100%. During the follow-up visits with all 101 subjects, no patient needed further interventional treatment or surgery in the target vessel embolization segment or aneurysm. Such excellent clinical trial results strongly demonstrate the safety and efficacy of this product.
As of the announcement date, the Company has developed a total of 57 products and product candidates, including 34 products approved in China, eight products approved in Europe and 1 product approved in Brazil. Zylox-Tonbridge has become one of the most competitive domestic vascular interventional medical device platforms with comprehensive product solutions. Looking forward, the company plans to continue developing innovative products with significant clinical value to provide patients with high-quality and affordable solutions.